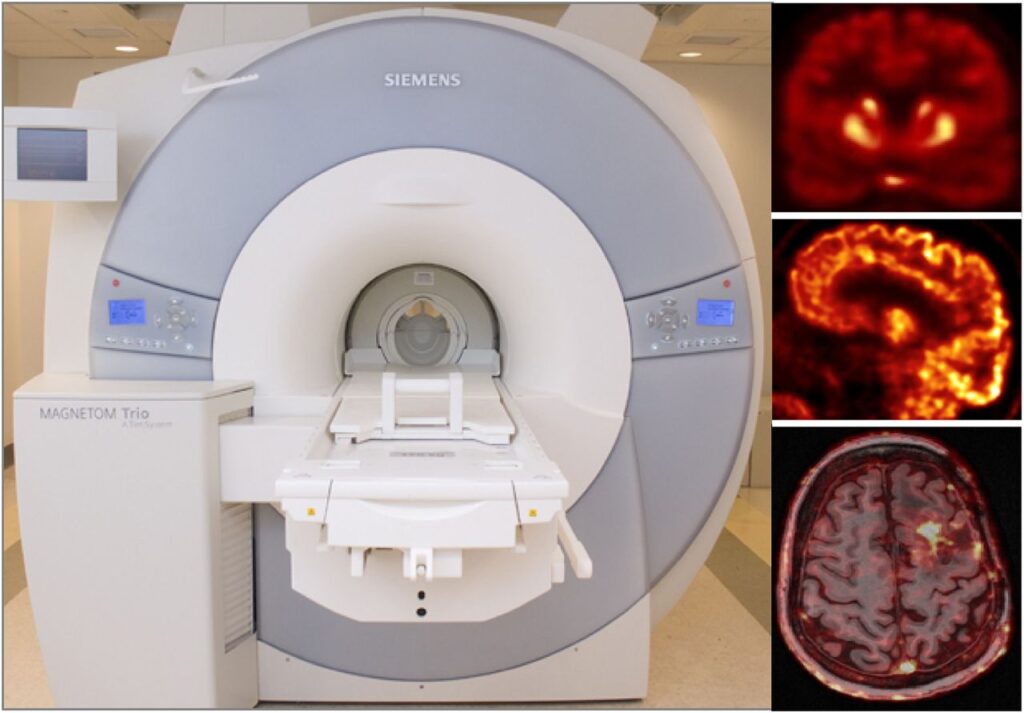
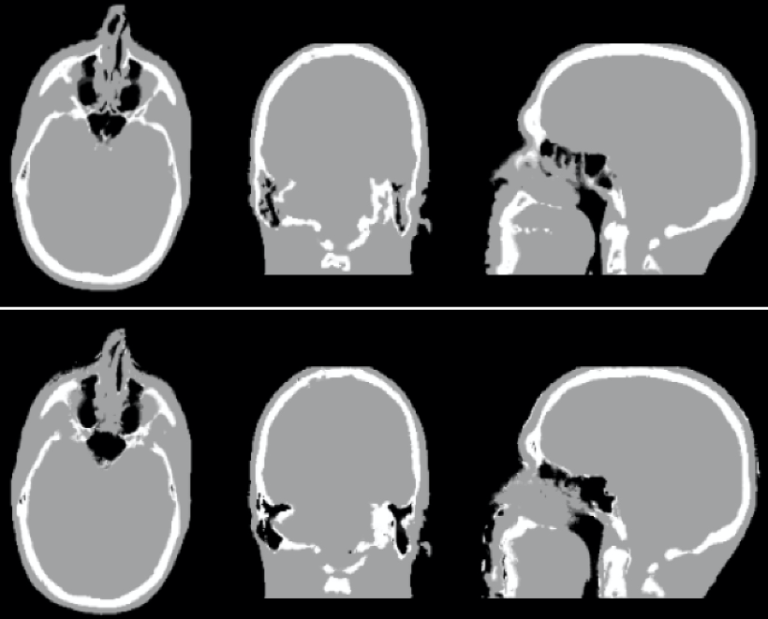
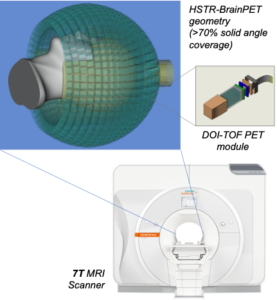
Partial volume effects lead in PET to an under-or overestimation of tissue activity concentration that depends on the activity distribution and the size and shape of the structures of interest, virtually affecting all neurological studies performed today. Although a myriad of partial volume effect correction methods has previously been proposed, no one method has been accepted or is routinely used for research applications and even less so in the clinical practice. We investigated the factors that affect the accuracy of these corrections:
- segmentation of structures of interest from the anatomical MR data;
- spatial co-registration of the MR and PET volumes;
- characterization of the scanner’s point spread function and the assumptions made during the correction (Bowen 2013).
Anatomically-aided PET reconstruction
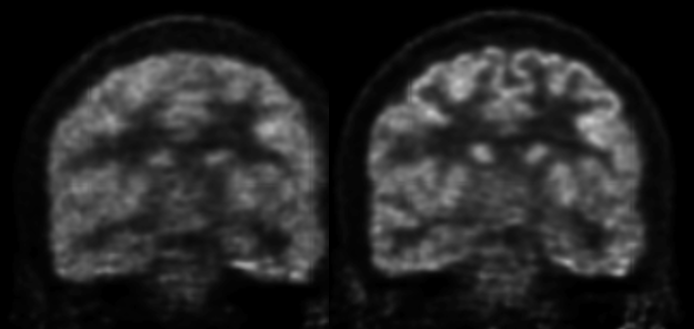
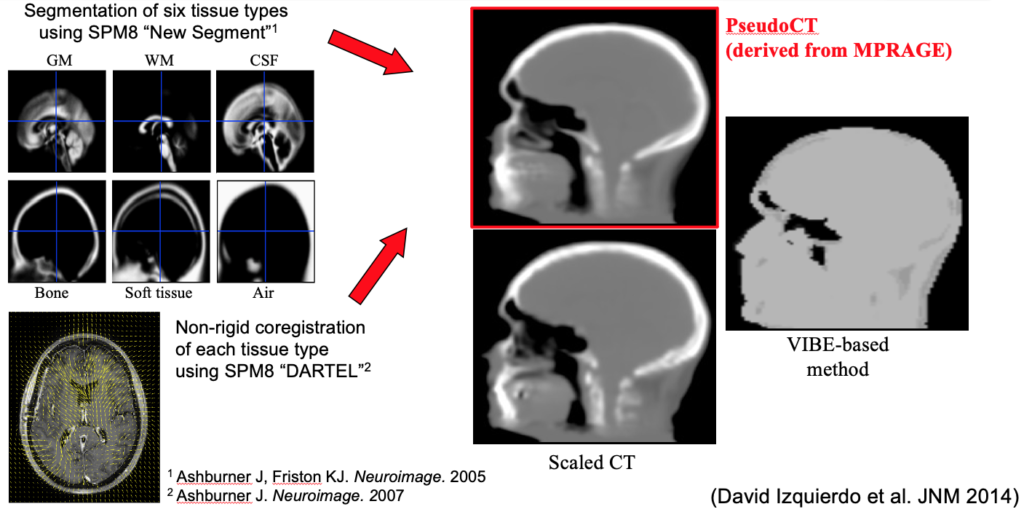
As a more elegant way to address this issue, in collaboration with Jinyi Qi’s group from the University of California at Davis, we have incorporated anatomical priors derived from high-resolution MRI into the PET image reconstruction model (Hutchcroft 2016).
MR-assisted PET Data Optimization in Simultaneous PET/MR
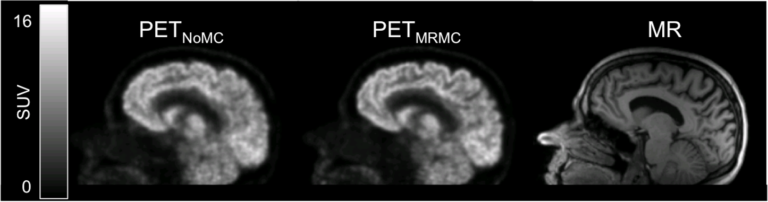
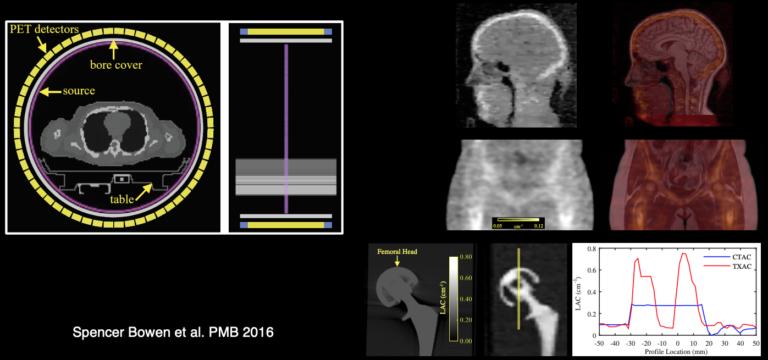
In addition to developing these individual methods, a major focus in our group has been on developing a unified data processing pipeline for integrating all these tools with the goal of improving the PET data quantification. We proposed an efficient algorithm to derive all the information required for performing:
- head attenuation and motion corrections,
- anatomy-aided reconstruction, and
- region-based analysis
from the standard data acquired in ~6 minutes, using one morphological MR sequence with embedded motion navigators (Chen 2019).
Using this approach, reduced variability and increased signal-to-noise ratio were seen after motion correction and anatomy-aided reconstruction. These results suggested PET data optimization may enable a more careful assessment of subtle changes in brain metabolism and allow for reduced sample sizes in future clinical trials.