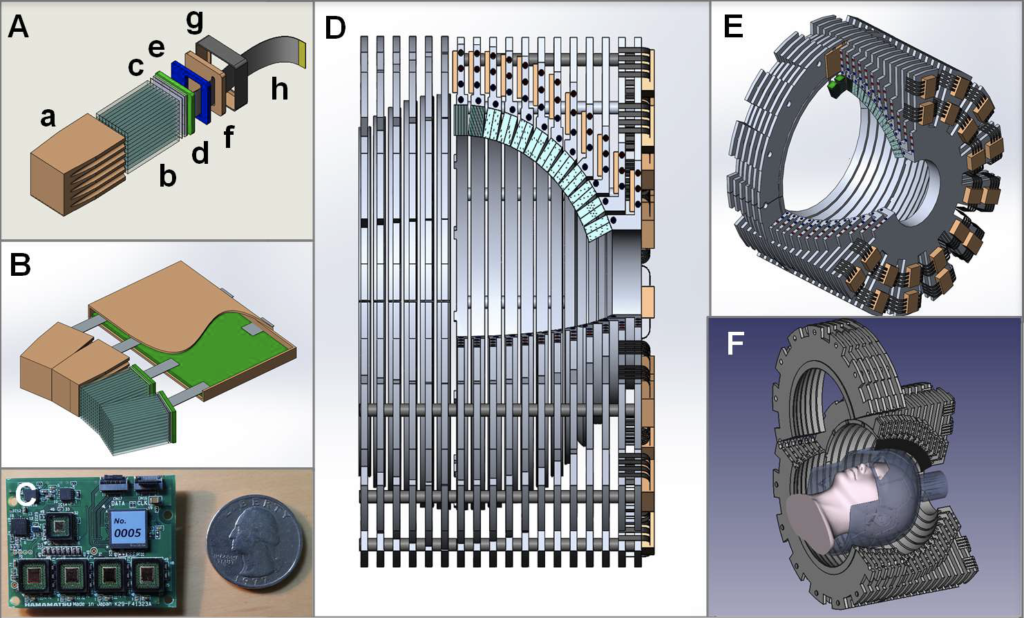
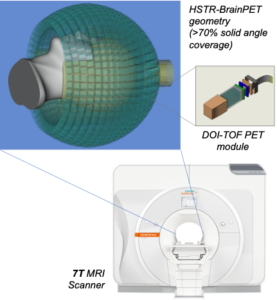
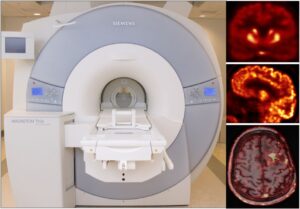
Attenuation correction for pelvis PET-MR imaging
The pelvis is another area where MR-based approaches that do not properly account for bone attenuation could lead to substantial bias in PET data quantification and deep learning approaches have been proposed to minimize this bias starting from the MR data acquired using specialized MR sequences. Instead, we implemented a deep learning-based approach to generate pseudo-CT maps exclusively from the Dixon-VIBE MR images routinely acquired for attenuation correction on the Biograph mMR scanner. Using our 2D network that takes four contrasts as inputs (water, fat, in-phase, and out-of-phase Dixon-VIBE images), the mean absolute relative change in PET values in the pelvis area was 2.36% ± 3.15% (Torrado-Carvajal et al 2019).
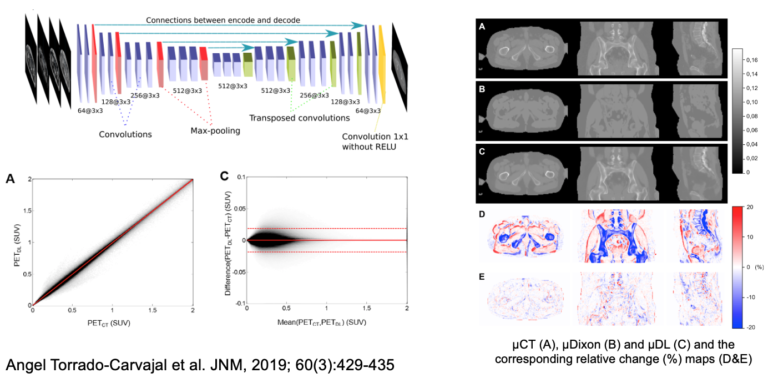
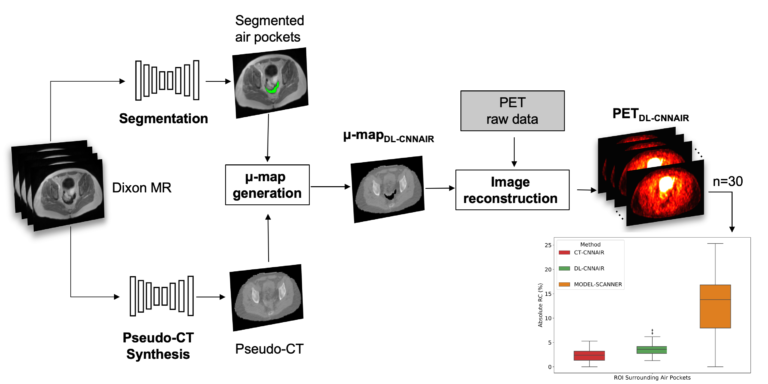
One remaining challenge in using convolutional neuronal networks to synthesize CT from MR images of the pelvis is the presence of air pockets (i.e., digestive tract gas) in this area. As CT and MR images are acquired on separate scanners at different times, the locations and sizes of these air pockets can change between the two scans, which can lead to errors in both the MR-CT co-registration and image synthesis tasks. We trained and evaluated CNNs to automatically segment air pockets from MR CAIPIRINHA-accelerated Dixon images and assessed the quantitative impact on the reconstructed PET images (Sari et al, submitted to JNM).