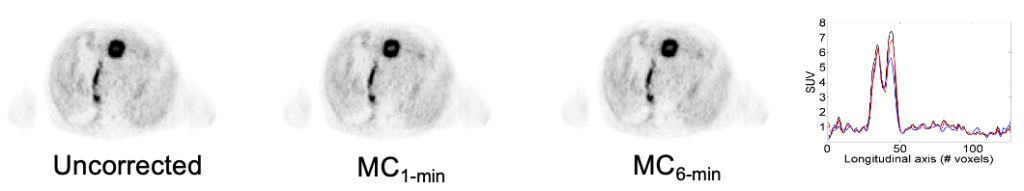
In the brain, kinetic modeling is frequently employed to study neuroreceptor properties and behavior. The binding potential is one such property, reflecting the density of available receptors and their affinity for the PET ligand. It can be measured by fitting a kinetic model to the PET time-activity curve with the additional input of activity concentration in arterial blood. When arterial blood is not collected as part of the study design, the kinetic model may instead be fit using an image-derived reference region that is devoid of specific binding.
However, the kinetic model selected can introduce bias into the measurements of interest. In the context of a behavioral-task study of dopamine release in healthy volunteers, we have shown that the use of MRTM2 (one of the most common reference tissue models) introduces bias into estimates change in binding potential. (Levine 2019 – abstract)