- Home
- >
- Image reconstruction







Partial volume effects lead in PET to an under-or overestimation of tissue activity concentration that depends on the activity distribution and the size and shape of the structures of interest, virtually affecting all neurological studies performed today. Although a myriad of partial volume effect correction methods has previously been proposed, no one method has been accepted or is routinely used for research applications and even less so in the clinical practice. We investigated the factors that affect the accuracy of these corrections:
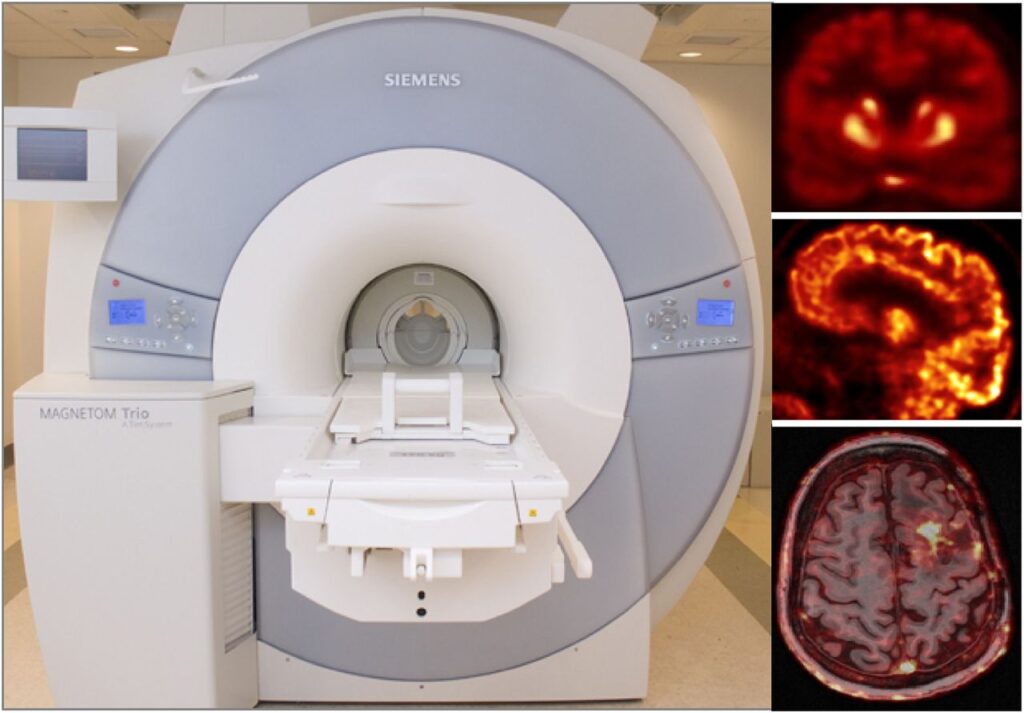
As a more elegant way to address this issue, in collaboration with Jinyi Qi’s group from the University of California at Davis, we have incorporated anatomical priors derived from high-resolution MRI into the PET image reconstruction model (Hutchcroft 2016).
In addition to developing these individual methods, a major focus in our group has been on developing a unified data processing pipeline for integrating all these tools with the goal of improving the PET data quantification. We proposed an efficient algorithm to derive all the information required for performing:
from the standard data acquired in ~6 minutes, using one morphological MR sequence with embedded motion navigators (Chen 2019).
Using this approach, reduced variability and increased signal-to-noise ratio were seen after motion correction and anatomy-aided reconstruction. These results suggested PET data optimization may enable a more careful assessment of subtle changes in brain metabolism and allow for reduced sample sizes in future clinical trials.

In the brain, kinetic modeling is frequently employed to study neuroreceptor properties and behavior. The binding potential is one such property, reflecting the density of available receptors and their affinity for the PET ligand. It can be measured by fitting a kinetic model to the PET time-activity curve with the additional input of activity concentration in arterial blood. When arterial blood is not collected as part of the study design, the kinetic model may instead be fit using an image-derived reference region that is devoid of specific binding.
However, the kinetic model selected can introduce bias into the measurements of interest. In the context of a behavioral-task study of dopamine release in healthy volunteers, we have shown that the use of MRTM2 (one of the most common reference tissue models) introduces bias into estimates change in binding potential. (Levine 2019 – abstract)

PET and MRI are two powerful imaging technologies that are characterized by high sensitivity and the ability to provide superior anatomic detail, respectively, which might make them ideal for evaluating the upper abdomen. However, PET requires long acquisition times, including the acquisition of data from moving organs, which may result in image blurring. On the other hand, MRI, especially standard DCE-MRI, can scan the chosen field of view in a shorter time, but requires the patient’s cooperation with the respiratory instructions and the ability to suspend respiration for the acquisition time of breath-hold sequences, usually in the range 14–20 s. Moreover, even in patients with an adequate respiratory breath-hold ability, the quality and the diagnostic information of DCE-MRI are also dependent on the hemodynamics of the patient and the timing of contrast agent injection and data acquisition. These variables explain the occurrence of respiratory artifacts and erroneous phases of contrast enhancement imaging in DCE-MRI.
After developing a novel self-navigated method that simultaneously corrects both PET data and DCE-MRI data for respiratory motion without increasing acquisition times, we focused on investigating its clinical performance and to compare the motion-corrected (MoCo) and uncorrected (non-MoCo) PET, MRI and fused PET/MRI data (Catalano 2018).
In this study, the quality of MoCo PET images was found to be higher than that of non-MoCo PET images. The improved quantitation allowed by MoCo PET might be clinically relevant. In fact, besides the contribution of PET quantitation to discriminating benign from malignant lesions, assessment of treatment response relies on measured differences in SUVmax and MTV along with possible complete visual disappearance of metabolically active lesions.
Furthermore, MoCo DCE-MRI might improve the overall quality of the data in patients unable to follow breathing instructions (deaf patients, patients with a different first language, or patients unable to hear the voice of the technologist while gradients are on), and in those with hemodynamic compromise in whom timing of arterial and portal venous phase imaging is challenging. The higher quality provided by fused PET/MRI data after MoCo has important clinical implications, as in the case of small but metabolically active lesions whose anatomic correlate might be difficult to identify. This is especially true when metabolic PET data are improperly fused over the MR anatomic overlay.

PET and MRI are two powerful imaging technologies that are characterized by high sensitivity and the ability to provide superior anatomic detail, respectively, which might make them ideal for evaluating the upper abdomen. However, PET requires long acquisition times, including the acquisition of data from moving organs, which may result in image blurring. On the other hand, MRI, especially standard DCE-MRI, can scan the chosen field of view in a shorter time, but requires the patient’s cooperation with the respiratory instructions and the ability to suspend respiration for the acquisition time of breath-hold sequences, usually in the range 14–20 s. Moreover, even in patients with an adequate respiratory breath-hold ability, the quality and the diagnostic information of DCE-MRI are also dependent on the hemodynamics of the patient and the timing of contrast agent injection and data acquisition. These variables explain the occurrence of respiratory artifacts and erroneous phases of contrast enhancement imaging in DCE-MRI.
We presented and evaluated in vivo a comprehensive approach for self-gated MR motion modeling applied to concurrent respiratory motion compensation of PET and DCE-MRI data acquired simultaneously in an integrated PET/MR system.
Fully registered, motion-corrected PET images and diagnostic DCE-MR images were obtained with negligible acquisition time prolongation compared with standard breath-hold techniques. Both the MR and the PET image quality and tracer uptake quantification were improved when compared with conventional methods (Fuin 2018).
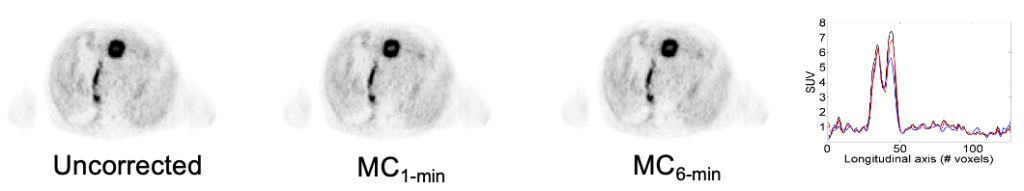
This approach was subsequently evaluated clinically in collaboration with Dr. Onofrio Catalano to demonstrate that motion-corrected PET/MRI produced better PET images and reduced the spatial mismatch between the two modalities (Catalano 2018).